Antimicrobial activity of crude henna extract against Gram-positive bacteria
Abstract
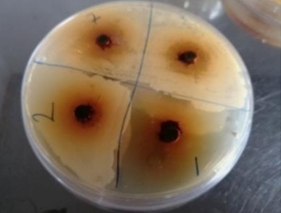
Henna plant has been used in herbal medicine for ages, but the medical uses of this plant as antimicrobial agent had not been well discussed. In this study we aim to examine the effect of ethanolic extract of local Basra henna leaves on Gram-positive bacteria species. Also, to assess the antibacterial properties of henna crude extract in vitro and compare them with antibiotics. Lawsonia inermis (henna) leaves were extracted with ethanol via using the solvent extraction technique. The pathogens were isolated from wound samples obtained from hospitalized patients in two different hospitals in Duhok city. Culture of thirty isolates had been recognized by routine methods. Different concentrations of ethanol crude extract were acquired and bio-assayed in vitro to inhibit the growth of five human pathogenic Gram-positive bacteria. Agar well diffusion assay was used for achieving henna antibiotic activity. Moreover, antibiotics susceptibility test was done by the disk diffusion method using Muller-Hinton agar medium. The growth of all tested bacteria was suppressed to various degrees by increasing the concentration of the extract. The data has revealed that Staphylococcus aureus was more sensitive than other examined isolates, where the diameter zone of inhibition were ranging from 16-27, 14-25, and 8-18 mm for Staphylococcus epidermidis, Lactobacillus spp. and Streptococcus pneumoniae respectively. The antimicrobial activity of henna extract indicates that it is suitable for being used as significant certain medications. Consequently, henna is active to serve as an anti-bacterial agent against multi-drug resistant Gram-positive bacteria.
References
2. Masoumian M, Zandi M. Antimicrobial activity of some medicinal plant extracts against multidrug resistant bacteria. Zahedan J Res Med Sci. 2017; 19(11): e10080.
3. Jastaniah S. The antimicrobial activity of some plant extracts, commonly used by Saudi people, against multidrug resistant bacteria. Life Sci J. 2014; 11(8): 78-84.
4. Mills-Robertson F, Onyeka C, Tay S, Walana W. In vitro antimicrobial activity of Antibact, an herbal medicinal product against standard and clinical bacterial isolates. J Med Plants Res. 2015; 9(11): 370-378.
5. Akter A, Neela F, Khan M, Islam M, Alam M. Screening of ethanol, petroleum ether and chloroform extracts of medicinal plants, Lawsonia inermis L. and Mimosa pudica L. for antibacterial activity. Indian J Pharmac Sci. 2010; 72(3): 388.
6. Raja W, Ovais M, Dubey A. Phytochemical screening and antibacterial activity of Lawsonia inermis leaf extract. Int J Microbiol Res. 2013; 4 (1): 33-36.
7. Natarajan V, Venugopal P, Menon T. Effect of Azadarichta indica (neem) on the growth pattern of dermatophtyes. Indian J Med Microbiol. 2003; 21: 98-101.
8. Misra S, Sahu K. Screening of some indigenous plants for antifungal activity against dermatophytes. Indian J Pharmacol. 1997; 9: 269-272.
9. Goyal B, Goyal R, Mehta A. Phyto-pharmacology of Achyranthes aspera: A review. Pharm Rev. 2007; 1: 143-153.
10. Uma Devi P, Murugan S, Suja S, Selvi S, Chinnaswamy P, Vijayanand E. Antibacterial, in vitro lipid per oxidation and phytochemical observation on Achyranthes bidentata Blume. Pak J Nutr. 2007; 6: 447-451.
11. Mohammed M, Ramadhan O, Hamoshy R. Study of the biological activity of compounds isolated from Lawsonia inermis. National J. 2006; 21: 112.
12. Amer S, Aly MM, Sabbagh S. Biocontrol of dermatophytes using some plant extracts and actinomycetes filtrates. Egypt J Biotech. 2007; 14: 291-315.
13. Mahmoud Y, Ebrahium M, Aly M. Influence of some plant extracts and microbioagents on some physiological traits of faba bean infected with Botrytis faba. Turkish J Bot. 2004; 7: 21-30.
14. Al-Rubiay K, Jaber N, Al-Mhaawe B, Alrubaiy L. Antimicrobial efficacy of henna extracts. Oman Med J. 2008; 23(4): 253.
15. Singh M, Jindal S, Kavia Z, Jangid B, Khem C. Traditional methods of cultivation and processing of henna. Henna, cultivation, improvement and trade. 2005; 21-34.
16. Ali K, Al-hood F, Obad K, Alshakka M. Phytochemical screening and antibacterial activity of Yemeni Henna (Lawsonia inermis) against some bacterial pathogens. J Pharm Biol Sci. 2016; 11: 24-27.
17. Hemem S. Activity of some plant extracts against common pathogens in bacterial skin infection: thesis MSc, College of Education, Basra University, Iraq, 2002.
18. Habbal O, Hasson S, El-Hag A, Al-Mahrooqi Z, Al-Hashmi N, Al-Bimani Z, et al. Antibacterial activity of Lawsonia inermis Linn (Henna) against Pseudomonas aeruginosa. Asian Pacif J Trop Biomed. 2011; 1(3): 173-176.
19. Body Shop. The Body Shop book of wellbeing: mind, body, soul. Ebury Press London. 1998: 173-192.
20. Sanni S, Thilza I, Ahmed M, Sanni F, Muhammed T, Okwor G. The effect of aqueous leaves extract of henna (Lawsonia inermis) in carbon tetrachloride induced hepato-toxicity in swiss albino mice. Acad Arena. 2010; 2(6): 87-89.
21. Harborne, Baxter H, Moss G. A handbook of bioactive compounds from plants. Phytochemical dictionary. 1995.
22. Dasgupta T, Rao A, Yadava P. Modulatory effect of henna leaf (Lawsonia inermis) on drug metabolising phase I and phase II enzymes, antioxidant enzymes, lipid peroxidation and chemically induced skin and forestomach papillomagenesis in mice. Mol Cell Biochem. 2003; 245: 11-22.
23. Ali B, Bashir A, Tanira M. Anti-inflammatory, antipyretic and analgesic effects of Lawsonia inermis L (Henna) in rats. Pharmacology. 1995; 51: 356-363.
24. Borade A, Kale B, Shete R. A phytopharmacological review on Lawsonia inermis (Linn.). Int J Pharm Life Sci. 2011; 2(1): 536-541.
25. Dahake P, Kamble S. Study on antimicrobial potential and preliminary phytochemical screening of Lawsonia inermis Linn. Int J Pharmac Sci Res. 2015; 6(8): 3344.
26. Hema R, Kumaravel S, Gomathi S, Sivasubramaniam C. Gas Chromatography-mass spectroscopic analysis of Lawsonia inermis leaves. New York Sci J. 2010: 3(11): 141-143.
27. Habbal O, Al-Jabri A, El-Hag A, Al-Mahrooqi Z, Al-Hashmi N. In-vitro antimicrobial activity of Lawsonia inermis Linn (henna). A pilot study on the Omani henna. Saudi Med J. 2005; 26(1): 69-72.
28. Bauer A, Kirby W, Sherris J, Turck M. Antibiotic susceptibility testing by a standardised single disk method. Am J Clin Path. 1966; 45: 493-496.
29. Khalaphallah R, Soliman W. Effect of henna and roselle extracts on pathogenic bacteria. Asian Pac J Trop Dis. 2014; 4(4): 292-296.
30. Aljamali N. Study effect of medical plant extracts in comparison with antibiotic against bacteria. J Sci Innov Res. 2013; 2: 843-845.
31. Yaouba A, Tchikoua R, Tatsadjieu N. Antibacterial effect of plant extracts against some pathogenic bacteria. Int J Nat Prod Res. 2012; 1: 83-87.
32. Jothiprakasam V, Ramesh S, Rajasekharan S. Preliminary phytochemical screening and antibacterial activity of Lawsonia inermis Linn (Henna) leaf extracts against reference bacterial strains and clinically important AMPC beta-lactamases producing Proteus mirabilis. Int J Pharm Pharm Sci. 2013; 5(1): 219-222.
33. Rout G, Das S, Samontoray P, Das P. In-vitro micropropagation of Lawsonia inermis (Lythraceae). Int J Trop Bisl Conserv. 2001; 49: 1-7.
34. Elmanama A, Alyazji A, Abu-Gheneima N. Antibacterial, antifungal and synergistic effect of Lawsonia inermis, Punica granatum and Hibiscus sabdariffa. Ann Alquds Med. 2011; 7: 33-41.
35. Muhammad H, Muhammad S. The use of Lawsonia inermis Linn. (Henna) in the management of burn wound infection. Afr J Biotechnol. 2005; 4(9): 934-937.
36. Bhuvaneswari K, Poongothai S, Kuruvilla A, Raju B. Inhibitory concentrations of Lawsonia inermis dry powder for urinary pathogens. Indian J Pharmacol. 2002; 34: 260-263.
37. Papageorgiou V, Assimopoulou A, Couladouros E, Hepworth D, Nicolaou K. The chemistry and biology of alkannin, shikonin, and related naphthazarin natural products. Angewandte Chemie. 1999; 38(3): 270-301.
38. Funke I, Melzig M. Traditionally used plants in diabetes therapy: phytotherapeutics as inhibitors of alpha-amylase activity. Rev Brasil Farmacogn. 2006; 16(1): 1-5.
39. Rahiman F, Mahmad N, Taha R, Elias H, Zaman F. Antimicrobial properties of Lawsonia inermis syn. Lawsonia alba in vivo and in vitro. J Food Agric Environ. 2013;11:502-504.
40. Hemem S. Activity of some plant extracts against common pathogens in bacterial skin infection. Thesis MSc, College of Education, Basra University, Iraq. 2002.
41. Habbal O, Al-Jabri A, El-Hag A, Al-Mahrooqi Z, Al-Hashmi N. In-vitro antimicrobial activity of Lawsonia inermis Linn (henna). A pilot study on the Omani henna. Saudi Med J. 2005; 26(1): 69-72.
42. Sharif S, Ismaeil A, Ahmad AA. Synergistic effect of different plant extracts and antibiotics on some pathogenic bacteria. Sci J Univ Zakho. 2020; 8(1): 7-11.

This work is licensed under a Creative Commons Attribution 4.0 International License.



